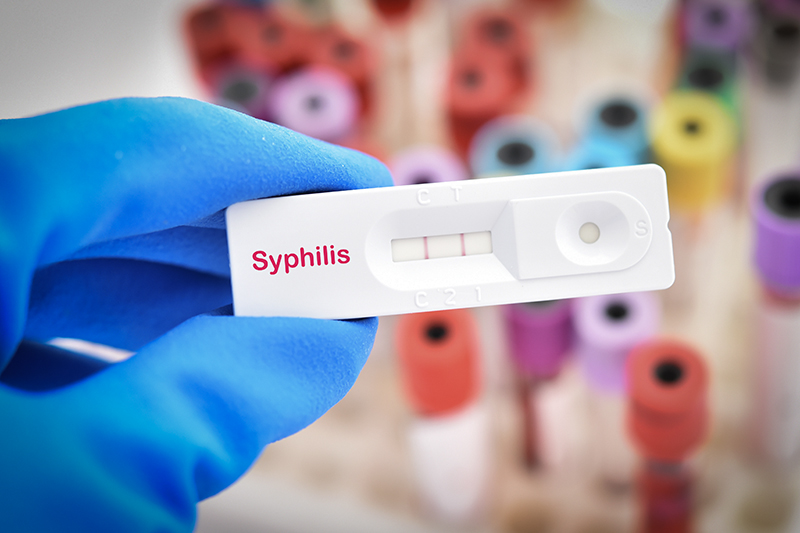
Mixed Messages on Blood
Advisory panel finds ban on gay blood donors ''suboptimal,'' but the way forward to remove it remains unclear
After a generation of gay men grew up being prohibited from donating blood after having had sex with just one man even one time since 1977, advocates were hopeful for a change. It’s 2010, after all, and even the trio of blood bank organizations, including the American Red Cross, opposes the ban that is euphemistically referred to as an ”indefinite deferral.”
Following the Health and Human Services Department’s Advisory Committee on Blood Safety and Availability meeting on June 10 and 11 to consider revising the longstanding policy this past week, though, it’s not yet any clearer whether the policy will be changing – and, if it does, when the change would happen. Despite that, for those gay men who wish to give blood, there are reasons to be hopeful.
On June 11, the advisory committee voted 6-9 against making a recommendation that the donation policy for men who have sex with men – or MSM – be changed now. Unfortunately, the question that was formally presented to the committee – ”Should the current indefinite deferral . . . be changed at the present time?” – appeared to have been interpreted in different ways by various committee members, leaving an unclear impression about what an affirmative vote would mean.

Because of this confusion – and after significant and substantive discussion – a few hours later the committee also voted 14-0 that the current deferral is ”suboptimal in permitting some high risk donations [from populations other than MSM] while preventing some low risk donations [from the MSM population].”
More significantly, they also recommended that HHS conduct the research necessary to evaluate the impact of ”modified eligibility criteria” for blood donation in both heterosexuals and homosexual populations and take the steps needed to validate that research.
No set timeline was put in the advisory committee’s recommendations, which will be considered by staff at HHS, including Assistant Secretary for Health Howard Koh, M.D. At that point, HHS officials could recommend changes be made by the Food and Drug Administration, which is charged by law with creating and enforcing standards for blood collection.
In the two-day meeting that preceded the votes, the advisory committee heard intensive testimony on topics ranging from the scientific rationale for the current policy to the reliability of donor questionnaires to gauge high-risk sexual behavior and heard the recommendations of representatives of Gay Men’s Health Crisis, AIDS Action and the Family Research Council.
In addition, the Red Cross, America’s Blood Centers and the AABB (formerly the American Association of Blood Banks) have – since 2006 – supported changing the policy to a one-year deferral, a position reiterated on June 11.
The Human Rights Campaign took a more broad-based approach in its comments to the committee, urging that the policy be changed to ”a risk-based assessment that will similarly defer gay and bisexual men who engage in high-risk behaviors [as with others who in engage in high-risk behaviors], while allowing those who are at low or no risk to contract HIV to donate.” In addition, Adam Bink at Open Left led a group of more than 15 LGBT and liberal blogs in a ”blogswarm” urging that readers send emails to HHS urging that the policy be revised.
The committee was presented by government officials with 5 possible options at the meeting, although it was explained that they were not limited to them: ”No change in current policy”; ”10 year deferral following last MSM exposure”; ”[f]ive year deferral following last MSM exposure”; ”[o]ne year deferral following last MSM exposure”; and ”[n]o specific deferral for MSM. Gender neutral questions based on specific activities.”
Unlike the blood banks, however, very few members of the advisory committee – overwhelmingly made up of doctors – were impressed with the idea of a one-year deferral policy. In fact, the members did not appear to be interested in any year-specific deferral policy, with committee members – most notably, Ileana Lopez-Plaza, M.D., the head of the transfusion medicine division at Henry Ford Health System in Detroit – urging that the committee focus more on general risk assessment than on what one member called ”arbitrary” deferral periods.
Committee member John Arnold, M.D., the president of Ohio Cardiac, Thoracic, and Vascular Surgeons, Inc., in Columbus, Ohio, criticized the ”very arbitrary timelines” set forward as possible changes, adding, ”We should go back to a risk-based deferral” for MSM donors.
Risk-based deferral, however, would involve changing the questionnaire used to screen potential donors for risk behaviors. As Harvey Klein, M.D., the head of the transfusion medicine division at the National Institutes of Health and a non-voting member of the advisory committee, explained, the questionnaire that has been carefully designed to ensure the most accurate responses from voluntary donors in order to screen out high-risk donors.
”Everyone could think why one or another question could be worded better,” Klein told the committee, ”but until you validate it” it’s not certain that it would be better.
Nonetheless, the risk-based assessment urged by HRC appeared to have the support of many of the committee members. As Lopez-Plaza said, the committee should have its aim to ”not fix a small part of it, but fix the big picture.” She later added, ”I want to look at this as [addressing] the entire donor population.”
After the initial vote, the committee members went into more detail discussing how they thought the process should move forward. Committee member Gregory J. Pomper, M.D., the blood bank director and a pathology professor at Wake Forest University School of Medicine, said of the first vote, ”The problem with making that determination today is that we don’t have an overlay” to address blood donation before criteria could be developed and validated for a non-deferral MSM donation policy. ”We really need to create a roadmap forward.”
Echoing Lopez-Plaza’s earlier comments, committee member Frederick B. Axelrod, M.D., representing America’s Blood Centers, suggested that the road map forward should look outside of simply MSM donation policies.
”There is a segment of the population that is prohibited from donating who does not provide a risk, and a segment that is allowed to donate but is at risk,” he said. ”There’s an opportunity to improve both at this time.”
NIH’s Klein expanded on that, addressing the possibility of changing the donor questionnaire and suggested that ”[r]esearch to look at the questions that could be asked should be recommended.”
Jay Epstein, M.D., the director of the Office of Blood Research and Review at the Food and Drug Administration and a non-voting member of the committee concurred, saying at one point, ”One research objective needs to be to attempt to validate low versus high risk questions not just for MSM but for heterosexual donors.”
In addition to concerns about the year-specific deferral policy change suggestion and the blood donor questionnaire, committee members discussed the possibility of combining some more limited deferral period – such as the one-year period – with mandatory HIV testing before donations would be taken while the blood donor questionnaire changes were being assessed.
Asked about the possibility of this pre-screening interim step, Nathan Schaefer, from GMHC, told the committee, ”If pre-screening were implemented as part of a temporary program, I think we would see that as a positive step.”
In the committee’s recommendations unanimously approved later in the afternoon on June 11, one of the tasks that they ask HHS to take is to ”determine the feasibility of donor pre-testing to limit risk while characterizing donors who might be recruited under modified eligibility criteria.”
Support Metro Weekly’s Journalism
These are challenging times for news organizations. And yet it’s crucial we stay active and provide vital resources and information to both our local readers and the world. So won’t you please take a moment and consider supporting Metro Weekly with a membership? For as little as $5 a month, you can help ensure Metro Weekly magazine and MetroWeekly.com remain free, viable resources as we provide the best, most diverse, culturally-resonant LGBTQ coverage in both the D.C. region and around the world. Memberships come with exclusive perks and discounts, your own personal digital delivery of each week’s magazine (and an archive), access to our Member's Lounge when it launches this fall, and exclusive members-only items like Metro Weekly Membership Mugs and Tote Bags! Check out all our membership levels here and please join us today!